Cardiac Safety
Every drug candidate filed with the FDA or EMA for clinical approval has to be evaluated for their potential to delay cardiac repolarization and cause heart arrhythmia.
We perform cardiac testing in two laboratories – our US-based lab for high- and medium-throughput testing of compounds in biochemical and cellular assay formats. Advanced studies are performed by our partner PharmaCore Labs covering compound testing on a broad cardiac ion channel panel under CiPA regulations as well as tissue- and organ-based studies. Together we cover all necessary cardiac tests during the pre-clinical evaluation of new drug candidates.
Cardiac liability testing is performed under non-GLP conditions for exploratory evaluation of new potential therapeutics only.
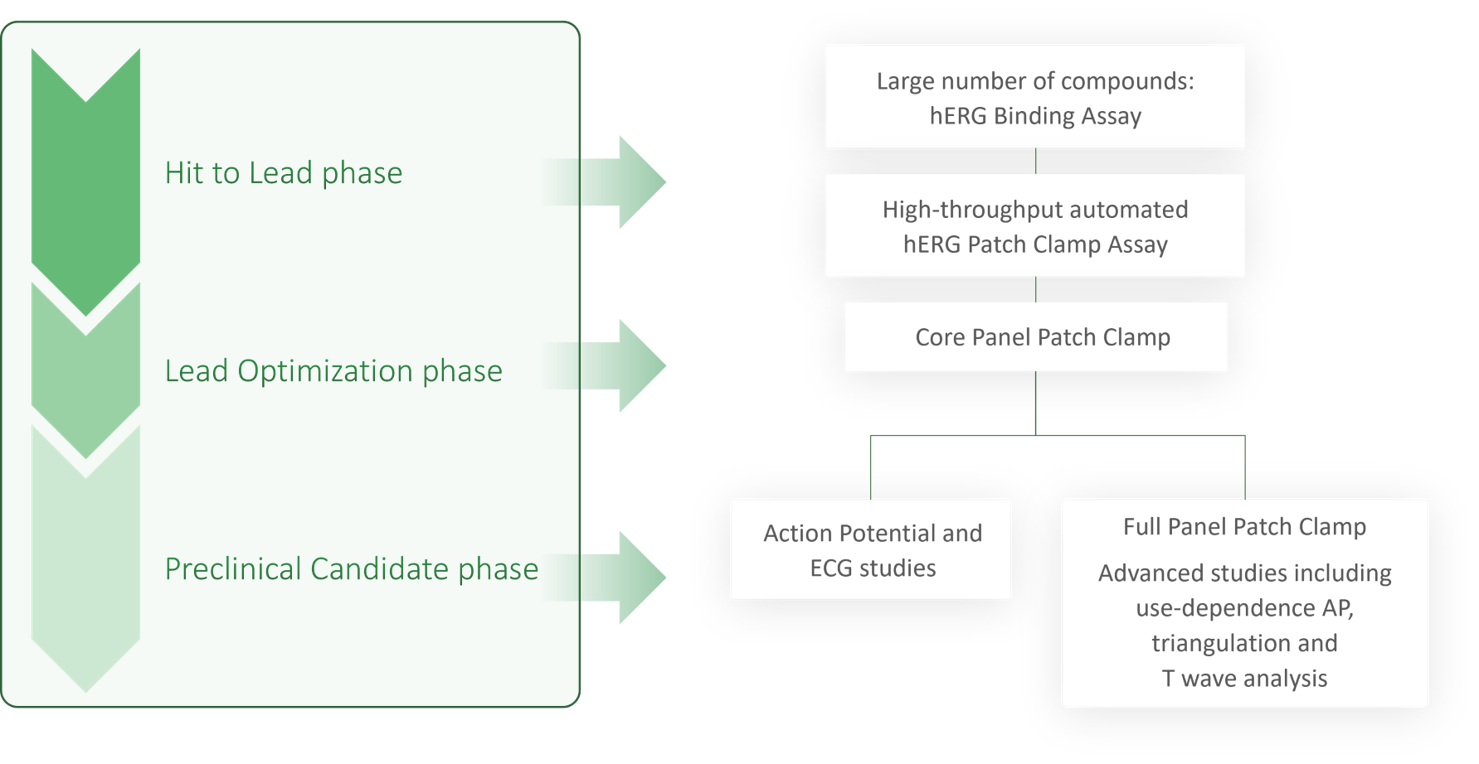
Cardiac liability assessment at Reaction Biology and Pharmacore Labs is performed at different steps during the drug discovery process.
- During hit-to-lead phase, first tests are performed in medium to high-throughput mode via biochemical binding studies of new compounds to hERG – the most frequently affected cardiac ion channel.
- Patch clamp studies on hERG expressing cell lines are part of the late hit-to-lead phase to discard unsuitable drug candidates early in the discovery process.
- Promising drug candidates are further screened on our core cardiac panel, including hERG, Nav1.5, and Cav1.2 via patch clamp during the lead optimization phase.
- Testing on the full cardiac ion channel panel is performed on cells via patch clamp following CiPA guidelines, including 6 or 7 ion channels. Further testing on cell culture can be conducted on human ventricular iPS (induced pluripotent stem cell)-derived monocytes via patch clamp to test the intracellular action potential.
- Promising lead candidates are tested in freshly isolated cardiac tissue such as Purkinje fibers, sinoatrial nodes, or papillary muscles for modulation of the intracellular action potential.
- ECG readings on freshly isolated beating rabbit hearts test effects on treatment during all phases of the heartbeat.
