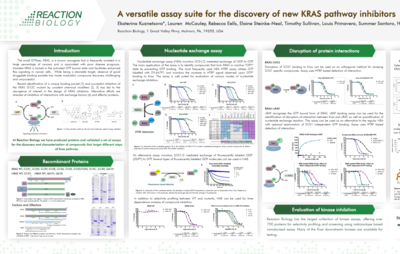
Thermal Shift Assay (Differential Scanning Fluorimetry) Service
Thermal shift assays utilize differential scanning fluorimetry (DSF) to measure the thermal stability of a target protein and the increase in protein melting temperature upon the binding of a stabilizing agent. It is useful for identifying ligands, buffer conditions, and cofactors affecting protein stability.
Thermal shift assays are performed via qPCR for high-throughput data acquisition in the 384 well format. We offer thermal shift assays for reader domain proteins, including bromodomains, chromodomains, and other methyl-readers. In addition, we offer custom-tailored thermal stability assay development for many other target categories.
Upon binding compounds cause a considerable change in the melting temperature of the target protein. These assays are carried out by our well-versed scientists with completion of standard studies within two weeks.
- The protein thermal shift assay is used to identify ligands that bind to the target, buffer conditions, and cofactors
- The differential scanning fluorimetry technique requires minimal assay establishment and is very cost-effective
- Deliverable: melting temperature shift (∆Tm) as a readout for compound binding affinity
Thermal shift assay uses the phenomenon in which ligand binding increases the target protein’s heat stability; this phenomenon may be utilized to gain insight in the compound-target interaction. Reach out to our business development team today to inquire about our differential scanning fluorimetry services.