CD47/SIRPα Pathway Targeting
Evaluate “don’t eat me” signal blockers that enhance macrophage phagocytosis of tumor cells; quantify ADCP when combined with tumor-targeting antibodies
Read our latest blog: LinkLight™ Protein-Protein Interaction AssaysLearn More
Profile macrophage polarization, phagocytosis, and inflammatory responses
Evaluate how therapeutic candidates modulate macrophage phenotype and function—from M1/M2 polarization dynamics to tumor cell phagocytosis and cytokine secretion.
Macrophages are highly plastic cells that adopt diverse functional states in response to environmental cues. Although the classical M1/M2 framework is a useful conceptual model, in vivo, macrophage phenotypes exist along a continuum of activation states.
M1-like macrophages, which are induced by IFN-γ or TLR ligands such as LPS, exhibit pro-inflammatory and tumoricidal activity. In contrast, M2-like macrophages, which are driven by IL-4, IL-13, or IL-10, display anti-inflammatory functions that can support tissue remodeling and tumor progression. In the tumor microenvironment, tumor-associated macrophages (TAMs) often exhibit immunosuppressive, M2-like characteristics that promote tumor growth, angiogenesis, and the suppression of anti-tumor immunity.
Therefore, targeting TAMs has emerged as a promising therapeutic strategy, including approaches that reprogram macrophages toward pro-inflammatory states or enhance their effector functions, such as CD40 agonists, STING pathway activation, and blockade of inhibitory signals like CD47.
Evaluate “don’t eat me” signal blockers that enhance macrophage phagocytosis of tumor cells; quantify ADCP when combined with tumor-targeting antibodies
Test STING agonists, TLR ligands, CD40 agonists, and CSF1R inhibitors for M2-to-M1 phenotype conversion
Measure how anti-PD-1/PD-L1 affects macrophage-mediated T cell suppression in co-culture systems
Assess immunomodulatory compounds for fibrosis, autoimmune, and inflammatory indications where macrophage dysfunction drives pathology
Combine surface marker profiling with functional readouts (phagocytosis, cytokine secretion, T cell suppression) for comprehensive mechanistic characterization.
Monocyte-derived macrophages from healthy donor PBMCs provide translational relevance that immortalized cell lines cannot match.
Flow cytometry for phenotyping, Cytation 5 for real-time imaging, MSD for cytokine multiplex—choose the readout that best fits your program.
Validated protocols for generating M0, M1, M2, TAM subtypes enable precise testing of polarization-modulating compounds.
Macrophage assays support therapeutic development in fibrosis, autoimmune disease, metabolic dysfunction, and infectious disease.
Test the effect of your immunotherapeutic compound on macrophage differentiation, within the tumour microenvironment or in response to inflammation and autoimmunity
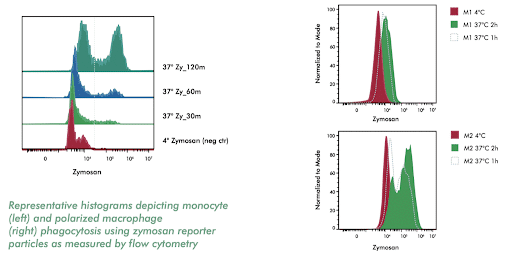
Assess the phagocytic capabilities of various monocyte/macrophage subtypes utilizing labeled bioparticles
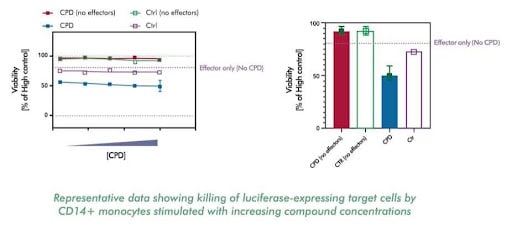
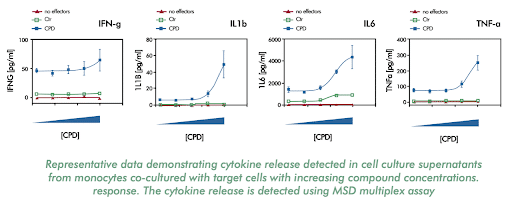
Investigate the efficacy of biologics by measuring their ability to kill target cells and induce monocytes to release inflammatory cytokines
Measure cytokines released upon differentiation or activation of immune response
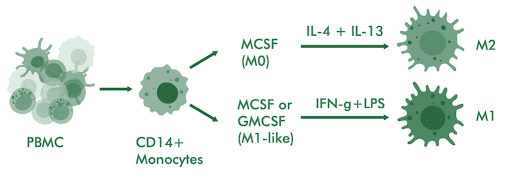
CD14+ monocytes are isolated from PBMCs and differentiated into M0 macrophages using M-CSF over 6-7 days. M1 polarization is induced with IFN-γ and LPS; M2 polarization uses IL-4 and IL-13 or IL-4, IL-10, or TGF-b depending on the desired subtype. Phenotype is confirmed by surface marker expression and cytokine profiling.
ADCC (antibody-dependent cellular cytotoxicity) involves NK cells killing antibody-coated targets. ADCP (antibody-dependent cellular phagocytosis) involves macrophages engulfing antibody-coated targets. Both are relevant for therapeutic antibodies; we offer assays for each mechanism.
We can generate TAM-like phenotypes using tumor-conditioned media or specific cytokine cocktails. Effects on TAM reprogramming can be measured through phenotype shifts and functional changes.
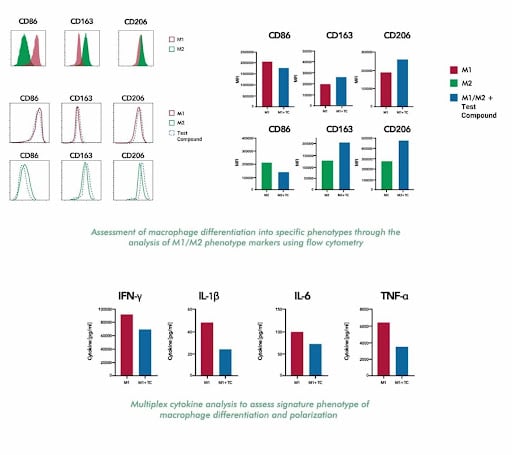
Surface marker shifts (decreased CD163/CD206, increased CD80/CD86), cytokine profile changes (decreased IL-10, increased IL-6/TNF-α/IL-12), and restored T cell activation in co-culture assays all indicate successful M1 reprogramming.
Absolutely. Macrophage polarization drives fibrosis (M2-like macrophages promote scarring), inflammatory diseases (M1-like drive tissue damage), and metabolic dysfunction. Our assays support therapeutic development across these indications.
Yes, we use fluorescently labeled tumor cell lines or standard bioparticles as targets. For tumor cell phagocytosis, we can combine with anti-CD47 or tumor-targeting antibodies to assess ADCP activity.
Evaluate T cell–mediated tumor cytotoxicity for checkpoint inhibitors and T cell engagers
Assess ADCC activity and NK cell–mediated killing for antibody therapeutics
Comprehensive cytokine and chemokine profiling via MSD platform
Evaluate TAM-targeting therapies in immunocompetent models with full immunophenotyping
From CD47 blockers to TAM reprogramming agents, our scientists will design a macrophage assay strategy aligned with your mechanism of action.