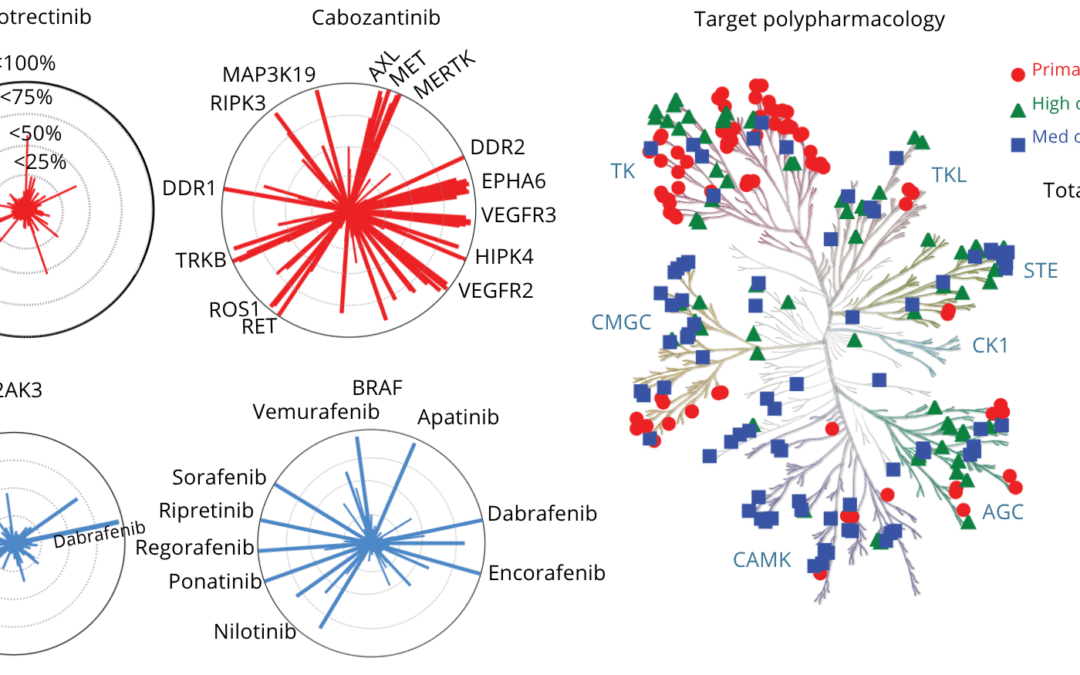
by Evelyn Ma | Mar 26, 2026 | Blog
Over 100 kinase inhibitors have now been approved by the FDA, transforming the treatment of cancer and other diseases.¹ Yet despite this progress, a key question remains underexplored: can these drugs be repurposed to target newly identified oncogenic kinase variants...
by Evelyn Ma | Feb 19, 2026 | Blog
Immunological impact across diseases Far more therapeutic areas involve immune regulation than traditional disease boundaries have suggested, leading to practical implications for how we profile drug candidates. Already a staple in immuno-oncology, autoimmune,...

by Evelyn Ma | Jan 26, 2026 | Blog
At Reaction Biology, quality is a commitment we can measure. We hold ourselves accountable for data integrity and report delivery metrics that define a quantifiable standard for our scientific operations. These performance metrics govern how we generate and deliver...
by Simon Clarke | Oct 13, 2025 | Blog
PPI biology balances promise and risk Protein-protein interactions (PPIs) are at the center of many critical cellular processes. They guide signaling, regulate gene expression, and ensure the smooth coordination of biological pathways. Dysregulation and disruption of...

by Simon Clarke | Feb 21, 2025 | Blog
One of the greatest challenges in oncology research is developing preclinical cancer models that accurately reflect human disease. The industry has long relied on subcutaneous tumor models for early-stage drug testing. These models, which involve injecting cultured...

by Andrew Khouri | Feb 11, 2025 | Blog
Adverse drug reactions (ADRs) remain one of the most significant challenges in drug development. Remarkably, around 75% of these ADRs are dose-dependent and can be predicted based on the pharmacological profiles of drug candidates. The only in vitro pharmacology assay...






