
by Evelyn Ma | May 14, 2026 | Newsletter
Headlines In This Issue Scientific Spotlight: Immuno-Oncology Explore how tumor-associated macrophages influence therapeutic outcomes and why comprehensive preclinical models are essential for IO drug development Featured Research: AACR 2026 Poster New data on...
by Evelyn Ma | Mar 26, 2026 | Blog
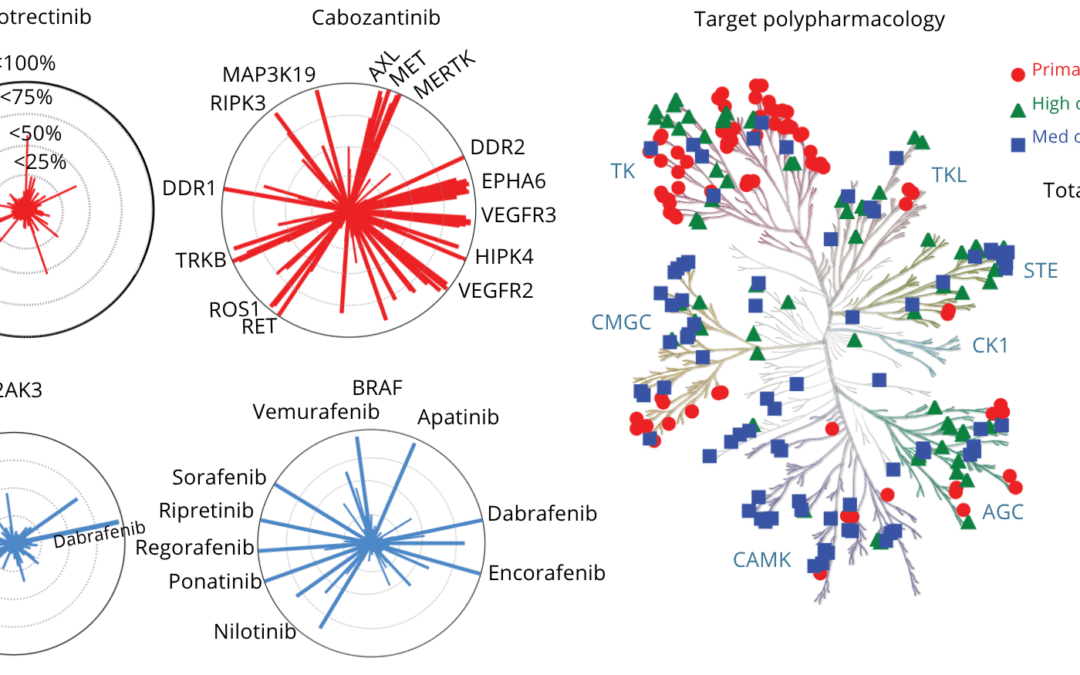
Over 100 kinase inhibitors have now been approved by the FDA, transforming the treatment of cancer and other diseases.¹ Yet despite this progress, a key question remains underexplored: can these drugs be repurposed to target newly identified oncogenic kinase variants...
by Evelyn Ma | Feb 19, 2026 | Blog
Immunological impact across diseases Far more therapeutic areas involve immune regulation than traditional disease boundaries have suggested, leading to practical implications for how we profile drug candidates. Already a staple in immuno-oncology, autoimmune,...

by Evelyn Ma | Jan 26, 2026 | Blog
At Reaction Biology, quality is a commitment we can measure. We hold ourselves accountable for data integrity and report delivery metrics that define a quantifiable standard for our scientific operations. These performance metrics govern how we generate and deliver...
by Evelyn Ma | Jan 19, 2026 | Newsletter
Headlines In This Issue Featured Research: Preclinical Models Our novel approach maintains BBB integrity in preclinical brain tumor models, enabling more predictive assessment of CNS therapeutic penetration and efficacy. Scientific Spotlight: GPCRs and...




