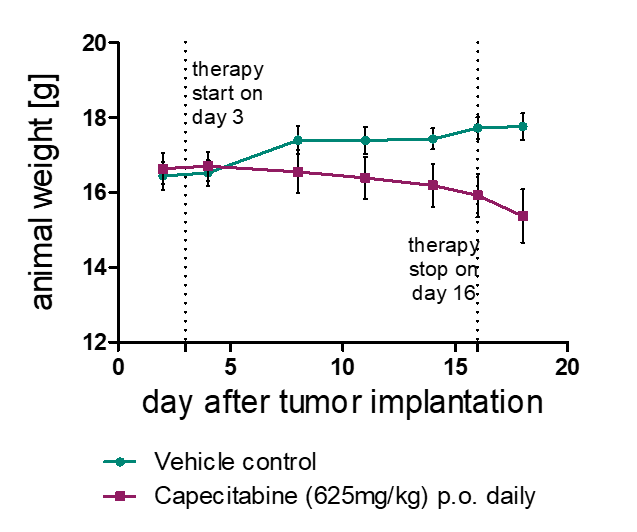
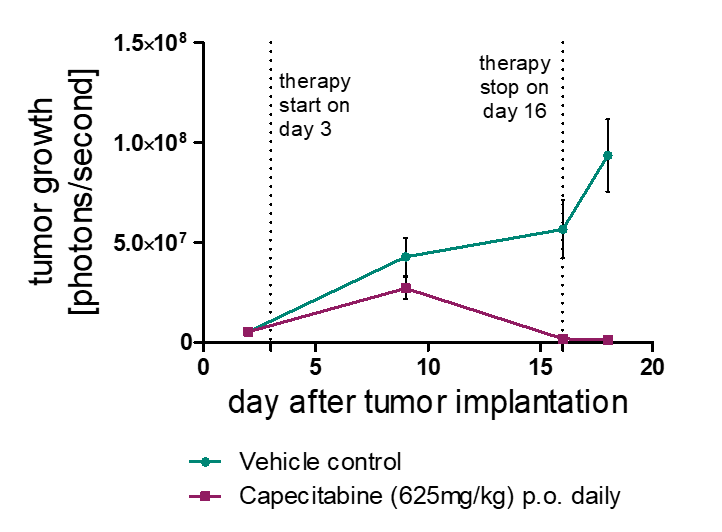Orthotopic Mouse Models
Tumors of orthotopic models grow in the organ of origin in their natural tumor environment. The interaction with stromal cells as well as tissue-specific immune cells allows tumor growth similar to that of the original tumor makes orthotopic tumor models very relevant for meaningful drug testing. Overall, orthotopic models feature a high predictive therapeutic value.
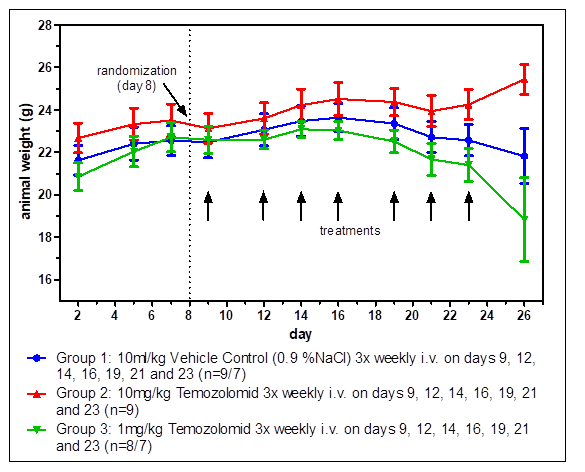
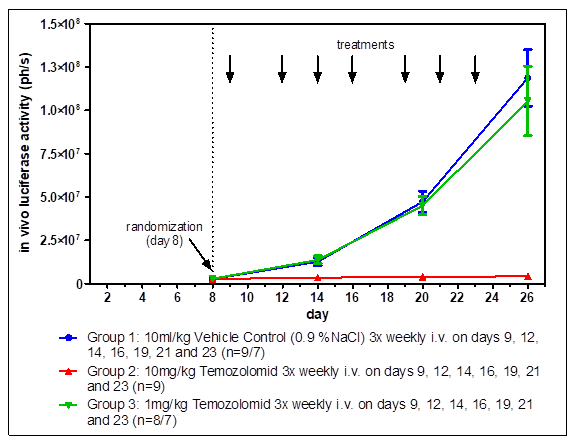
The interaction of tumor cells with tissue-specific stroma cells affects tumor growth, differentiation, and drug sensitivity. Moreover, orthotopic tumors can metastasize with specificities comparable to the human situation. Orthotopic implantation of tumors generally requires surgery and methods for visualizing tumor growth inside the body. Orthotopic imaging via our state-of-the-art IVIS bioimager yields in-depth knowledge about the growth characteristics of the tumors.
- Orthotopic tumors are implanted into the organ of origin
- Study results have high translational value
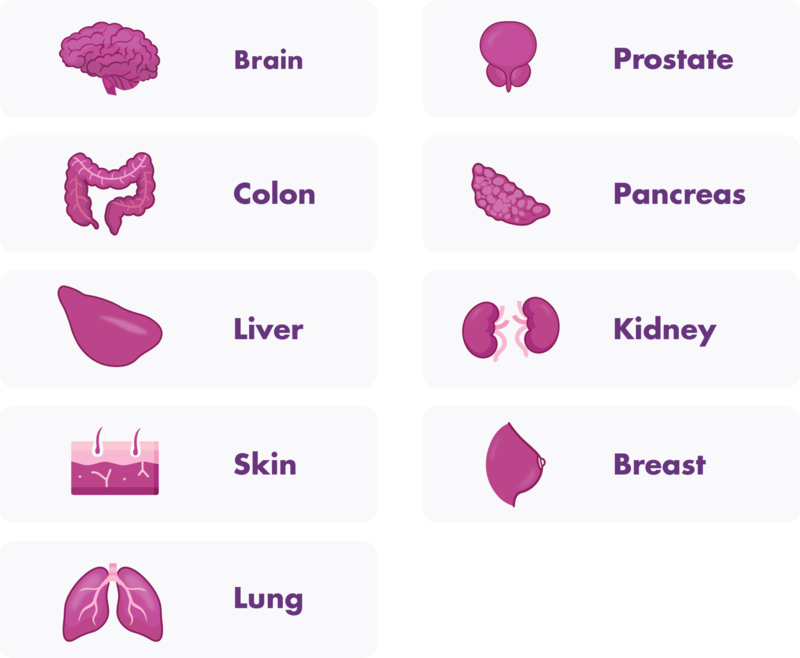
- The following syngeneic and xenograft orthotopic mouse models are available: Pancreatic tumor models, colon cancer models, breast cancer models, prostate cancer models, lung cancer models, skin cancer models such as melanoma, brain cancer models such as glioblastoma, and a kidney cancer model.
- Our veterinary surgeons have extensive experience and training in leak-free surgical techniques for orthotopic tumor implantation
- Orthotopic model reports are produced by PhD-level medical writers and are custom-tailored to each project
- Meticulous and standardized documentation process according to GV-Solas requirements and in line with ISO 9001 certification
Reaction Biology has a global network of business development managers to understand your specific research needs and ensure your needs are met. Throughout the project, you will work with just one team of scientists to facilitate consistent communication. If you have inquiries about our syngeneic or xenograft orthotopic models or if you want to request a quote, contact us today.